Given the recent news that biodiesel has caused buses in Minnesota to malfunction in cold weather, I thought this would be a good time to review the differences between diesel, biodiesel, and green diesel. In order to explain the key issues, I am going to excerpt from the chapter on renewable diesel that I wrote for Biofuels, Solar and Wind as Renewable Energy Systems: Benefits and Risks.
First, what happened in Minnesota?
Biodiesel fuel woes close Bloomington schools
All schools in the Bloomington School District will be closed today after state-required biodiesel fuel clogged in school buses Thursday morning and left dozens of students stranded in frigid weather, the district said late Thursday.
Rick Kaufman, the district’s spokesman, said elements in the biodiesel fuel that turn into a gel-like substance at temperatures below 10 degrees clogged about a dozen district buses Thursday morning. Some buses weren’t able to operate at all and others experienced problems while picking up students, he said.
And in case you think this was an isolated incident:
The decision to close school today came after district officials consulted with several neighboring districts that were experiencing similar problems. Bloomington staffers tried to get a waiver to bypass the state requirement and use pure diesel fuel, but they weren’t able to do so in enough time, Kaufman said. They also decided against scheduling a two-hour delay because the temperatures weren’t expected to rise enough that the problem would be eliminated.
What is Biodiesel?
Biodiesel is defined as the mono-alkyl ester product derived from lipid1 feedstock like SVO or animal fats (Knothe 2001). The chemical structure is distinctly different from petroleum diesel, and biodiesel has somewhat different physical and chemical properties from petroleum diesel.
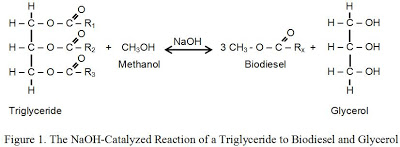
Biodiesel is normally produced by reacting triglycerides (long-chain fatty acids contained in the lipids) with an alcohol in a base-catalyzed reaction (Sheehan 1998) as shown in Figure 1. Methanol, ethanol, or even longer chain alcohols may be used as the alcohol, although lower-cost and faster-reacting methanol2 is typically preferred. The primary products of the reaction are the alkyl ester (e.g., methyl ester if methanol is used) and glycerol. The key advantage over straight vegetable oil (SVO) is that the viscosity is greatly reduced, albeit at the cost of additional processing and a glycerol byproduct.
The key thing to note here is that biodiesel contains oxygen atoms (the ‘O’ in the biodiesel structure above), but petroleum diesel and green diesel do not. This leads to different physical properties for biodiesel.
Biodiesel Characteristics
Biodiesel is reportedly nontoxic and biodegradable (Sheehan et al. 1998). An EPA study published in 2002 showed that the impact of biodiesel on exhaust emissions was mostly favorable (EPA 2002). Compared to petroleum diesel, a pure blend of biodiesel was estimated to increase the emission of NOx by 10%, but reduce emissions of carbon monoxide and particulate matter by almost 50%. Hydrocarbon emissions from biodiesel were reduced by almost 70% relative to petroleum diesel. However, other researchers have reached different conclusions. While confirming the NOx reduction observed in the EPA studies, Altin et al. determined that both biodiesel and SVO increase CO emissions over petroleum diesel (Altin et al. 2001). They also determined that the energy content of biodiesel and SVO was about 10% lower than for petroleum diesel. This means that a larger volume of biodiesel consumption is required per distance traveled, increasing the total emissions over what a comparison of the exhaust concentrations would imply.
The natural cetane3 number for biodiesel in the 2002 EPA study was found to be higher than for petroleum diesel (55 vs. 44). Altin et al. again reported a different result, finding that in most cases the natural cetane numbers were lower for biodiesel than for petroleum diesel. These discrepancies in cetane results have been attributed to the differences in the quality of the oil feedstock, and to whether the biodiesel had been distilled (Van Gerpen 1996).
A major attraction of biodiesel is that it is easy to produce. An individual with a minimal amount of equipment or expertise can learn to produce biodiesel. With the exception of SVO, production of renewable diesel by hobbyists is limited to biodiesel because a much larger capital expenditure is required for other renewable diesel technologies.
Biodiesel does have characteristics that make it problematic in cold weather conditions. The cloud and pour points4 of biodiesel can be 20° C or higher than for petroleum diesel (Kinast 2003). This is a severe disadvantage for the usage of biodiesel in cold climates, and limits the blending percentage with petroleum diesel in cold weather.
Green Diesel
Definition
Another form of renewable diesel is ‘green diesel.’ Green diesel is chemically the same as petroleum diesel, but it is made from recently living biomass. Unlike biodiesel, which is an ester and has different chemical properties from petroleum diesel, green diesel is composed of long-chain hydrocarbons, and can be mixed with petroleum diesel in any proportion for use as transportation fuel. Green diesel technology is frequently referred to as second-generation renewable diesel technology.
There are two methods of making green diesel. One is to hydroprocess vegetable oil or animal fats. Hydroprocessing may occur in the same facilities used to process petroleum. The second method of making green diesel involves partially combusting a biomass source to produce carbon monoxide and hydrogen – syngas – and then utilizing the Fischer-Tropsch reaction to produce complex hydrocarbons. This process is commonly called the biomass-to-liquids, or BTL process.
Hydroprocessing
Hydroprocessing is the process of reacting a feed stock with hydrogen under elevated temperature and pressure in order to change the chemical properties of the feed stock. The technology has long been used in the petroleum industry to ‘crack’, or convert very large organic molecules into smaller organic molecules, ranging from those suitable for liquid petroleum gas (LPG) applications through those suitable for use as distillate fuels.
In recent years, hydroprocessing technology has been used to convert lipid feed stocks into distillate fuels. The resulting products are a distillate fuel with properties very similar to petroleum diesel, and propane (Hodge 2006). The primary advantages over first-generation biodiesel technology are: 1). The cold weather properties are superior; 2). The propane byproduct is preferable over glycerol byproduct; 3). The heating content is greater; 4). The cetane number is greater; and 5). Capital costs and operating costs are lower (Arena et al. 2006).
A number of companies have announced renewable diesel projects based on hydroprocessing technology. In May 2007 Neste Oil Corporation in Finland inaugurated a plant that will produce 170,000 t/a of renewable diesel fuel from a mix of vegetable oil and animal fat (Neste 2007). Italy’s Eni has announced plans for a facility in Leghorn, Italy that will hydrotreat vegetable oil for supplying European markets. Brazil’s Petrobras is currently producing renewable diesel via their patented hydrocracking technology (NREL 2006). And in April 2007 ConocoPhillips, after testing their hydrocracking technology to make renewable diesel from rapeseed oil in Whitegate, Ireland, announced a partnership with Tyson Foods to convert waste animal fat into diesel (ConocoPhillips 2007).
Like biodiesel production, which normally utilizes fossil fuel-derived methanol, hydroprocessing requires fossil fuel-derived hydrogen5. No definitive life cycle analyses have been performed for diesel produced via hydroprocessing. Therefore, the energy return and overall environmental impact have yet to be quantified.
Biomass-to-Liquids
When an organic material is burned (e.g., natural gas, coal, biomass), it can be completely oxidized (gasified) to carbon dioxide and water, or it can be partially oxidized to carbon monoxide and hydrogen. The latter partial oxidation (POX), or gasification reaction, is accomplished by restricting the amount of oxygen during the combustion. The resulting mixture of carbon monoxide and hydrogen is called synthesis gas (syngas) and can be used as the starting material for a wide variety of organic compounds, including transportation fuels.
Syngas may be used to produce long-chain hydrocarbons via the Fischer-Tropsch (FT) reaction. The FT reaction, invented by German chemists Franz Fischer and Hans Tropsch in the 1920s, was used by Germany during World War II to produce synthetic fuels for their war effort. The FT reaction has received a great deal of interest lately because of the potential for converting natural gas, coal, or biomass into liquid transportation fuels. These processes are respectively referred to as gas-to-liquids (GTL), coal-to-liquids (CTL), and biomass-to-liquids (BTL), and the resulting fuels are ‘synthetic fuels’ or ‘XTL fuels’. Of the XTL processes, BTL produces the only renewable fuel, as it utilizes recently anthropogenic (atmospheric) carbon.
Renewable diesel produced via BTL technology has one substantial advantage over biodiesel and hydrocracking technologies: Any source of biomass may be converted via BTL. Biodiesel and hydrocracking processes are limited to lipids. This restricts their application to a feedstock that is very small in the context of the world’s available biomass. BTL is the only renewable diesel technology with the potential for converting a wide range of waste biomass.
Like GTL and CTL, development of BTL is presently hampered by high capital costs. According to the Energy Information Administration’s Annual Energy Outlook 2006, capital costs per daily barrel of production are $15,000-20,000 for a petroleum refinery, $20,000-$30,000 for an ethanol plant, $30,000 for GTL, $60,000 for CTL, and $120,000-$140,000 for BTL (EIA 2006).
While a great deal of research, development, and commercial experience has gone into FT technology in recent years6, biomass gasification biomass gasification technology is a relatively young field, which may partially explain the high capital costs. Nevertheless, the technology is progressing. Germany’s Choren is building a plant in Freiberg, Germany to produce 15,000 tons/yr of their SunDiesel® product starting in 2008 (Ledford 2006).
Straight Vegetable Oil
Unmodified vegetable-derived triglycerides, commonly known as vegetable oil, may also be used to fuel a diesel engine. Rudolf Diesel demonstrated the use of peanut oil as fuel for one of his diesel engines at the Paris Exposition in 1900 (Altin et al. 2001). Modern diesel engines are also capable of running on straight (unmodified) vegetable oil (SVO) or waste grease, with some loss of power over petroleum diesel (West 2004). Numerous engine performance and emission tests have been conducted with SVO derived from many different sources, either as a standalone fuel or as a mixture with petroleum diesel (Fort and Blumberg 1982, Schlick et al. 1988, Hemmerlein et al. 1991, Goering et al. 1982).
The advantage of SVO as fuel is that a minimal amount of processing is required, which lowers the production costs of the fuel. The energy return for SVO, defined as energy output over the energy required to produce the fuel, will also be higher due to the avoidance of energy intensive downstream processing steps.
There are several disadvantages of using SVO as fuel. The first is that researchers have found that engine performance suffers, and that hydrocarbon and carbon monoxide emissions increase relative to petroleum diesel. Particulate emissions were also observed to be higher with SVO. However, the same studies found that nitrogen oxide (NOx) emissions were lower for SVO (Altin et al. 2001). On long-term tests, carbon deposits have been found in the combustion chamber, and sticky gum deposits have occurred in the fuel lines (Fort and Blumberg 1982). SVO also has a very high viscosity relative to most diesel fuels. This reduces its ability to flow, especially in cold weather. This characteristic may be compensated for by heating up the SVO, or by blending it with larger volumes of lower viscosity diesel fuels.
Conclusions
In order to understand the potential problems with biodiesel under cold weather conditions, it is important to understand that biodiesel is chemically different from petroleum or green diesel – and thus should not be expected to have the same chemical properties. Biodiesel is an ester, while petroleum and green diesel are hydrocarbons. The only reason it is called ‘diesel’ is that it can fuel a diesel engine. Likewise vegetable oil, butanol, and even ethanol blends could be called ‘diesels’, as each of these can be used to fuel a diesel engine.
Finally, it should also be noted that petroleum diesel is not immune from cold weather gelling. It is just that these problems don’t begin to occur until the temperatures are much lower than those at which biodiesel begins to gel. If extremely cold weather conditions are likely, then petroleum diesel is blended differently. More kerosene is put into the mixture, which is a lighter diesel (and has a shorter carbon chain length and is just a little heavier than gasoline) and is referred to as #1 diesel.
Footnotes
1. Lipids are oils obtained from recently living biomass. Examples are soybean oil, rapeseed oil, palm oil, and animal fats. Petroleum is obtained from ancient biomass and will be specifically referred to as ‘crude oil’ or the corresponding product ‘petroleum diesel.’
2. Methanol is usually produced from natural gas, although some is commercially produced from light petroleum products or from coal. Methanol therefore represents a significant – but often overlooked – fossil fuel input into the biodiesel process.
3. The cetane number is a measure of the ignition quality of diesel fuel based on ignition delay in a compression ignition engine. The ignition delay is the time between the start of the injection and the ignition. Higher cetane numbers mean shorter ignition delays and better ignition quality.
4. The cloud point is the temperature at which the fuel becomes cloudy due to the precipitation of wax. The pour point is the lowest temperature at which the fuel will still freely flow.
5. Hydrogen is produced almost exclusively from natural gas.
6. Companies actively involved in developing Fischer-Tropsch technology include Shell, operating a GTL facility in Bintulu, Malaysia since 1993; Sasol, with CTL and GTL experience in South Africa; and ConocoPhillips and Syntroleum, both with GTL demonstration plants in Oklahoma.
References
Altin, R., Cetinkaya S., & Yucesu, H.S. (2001). The potential of using vegetable oil fuels as fuel for Diesel engines. Energy Convers. Manage. 42, 529–538. Arena, B.; Holmgren, J.; Marinangeli, R.; Marker, T.; McCall, M.; Petri, J.; Czernik, S.; Elliot, D.; & Shonnard, D. (2006, September). Opportunities for Biorenewables in Petroleum Refineries (Paper presented at the Rio Oil & Gas Expo and Conference, Instituto Braserileiro de Petroleo e Gas). ConocoPhillips. (2007). ConocoPhillips and Tyson Foods Announce Strategic Alliance To Produce Next Generation Renewable Diesel Fuel. Retrieved July 21, 2007 from the ConocoPhillips corporate web site: http://www.conocophillips.com/newsroom/news_releases/2007+News+Releases/041607.htm EIA, Energy Information Administration. (2006). Annual Energy Outlook 2006. DOE/EIA-0383, 57-58. EPA, U.S. Environmental Protection Agency. (2002). A Comprehensive Analysis of Biodiesel Impacts on Exhaust Emissions. EPA420-P-02-001. Fort, E. F. & Blumberg, P. N. (1982). Performance and durability of a turbocharged diesel fueled with cottonseed oil blends. (Paper presented at the International Conference on Plant and Vegetable Oils as Fuel, ASAE). Goering C.E., Schwab, A. Dougherty, M. Pryde, M. & Heakin, A. (1981). Fuel properties of eleven vegetable oils. (Paper presented at the American Society of Agricultural Engineers meeting, Chicago, IL, USA). Hodge, C. (2006). Chemistry and Emissions of NExBTL. (Presented at the University of California, Davis). Retrieved July 21, 2007 from http://bioenergy.ucdavis.edu/materials/NExBTL%20Enviro%20Benefits%20of%20paraffins.pdf Hemmerlein M., Korte V., & Richter HS. (1991). Performance, exhaust emission and durability of modern diesel engines running on rapeseed oil. SAE Paper 910848. Kinast, J. NREL, National Renewable Energy Laboratory. (2003). Production of Biodiesels from Multiple Feed-stocks and Properties of Biodiesels and Biodiesel/Diesel Blends. NREL/SR-510-31460. Knothe, G. (2001). Historical perspectives on vegetable oil-based diesel fuels. INFORM 12 (11), 1103–7. Ledford, H. (2006). Liquid fuel synthesis: Making it up as you go along. Nature 444, 677 – 678. Neste Oil Corporation. (2007). Neste Oil inaugurates new diesel line and biodiesel plant at Porvoo. Retrieved July 21, 2007 from http://www.nesteoil.com/default.asp?path=1,41,540,1259,1260,7439,8400 NREL, National Renewable Energy Laboratory. (2006). Biodiesel and Other Renewable Diesel Fuels, NREL/FS-510-40419 Sheehan, J. NREL, National Renewable Energy Laboratory. (1998). An Overview of Biodiesel and Petroleum Diesel Life Cycles, NREL/TP-580-24772. Schlick M. L., Hanna, M. A., & Schinstock, J. L. (1988). Soybean and sunflower oil performance in diesel engine. ASAE 31 (5). Van Gerpen, J. (1996). Cetane Number Testing of Biodiesel. (Paper presented at the Third Liquid Fuel Conference: Liquid Fuel and Industrial Products from Renewable Resources, St. Joseph, MI). West, T. (2004). The Vegetable-Oil Alternative. [Electronic version]. Car and Driver. Retrieved June 28, 2007 from http://www.caranddriver.com/article.asp?section_id=4&article_id=7818
Book Chapter Outline
While I have posted extended excerpts from my book chapter, I covered quite a bit more material in there. Here is the full chapter outline:
Renewable Diesel by Robert Rapier
1. The Diesel Engine 2. Ecological Limits 3. Straight Vegetable Oil (SVO) 4. Biodiesel 4.1.1. Definition/Production Process 4.1.2. Fuel Characteristics 4.1.3. Energy Return 4.1.4. Glycerin Byproduct 5. Green Diesel 5.1.1. Definition/Production 5.1.1.1. Hydroprocessing 5.1.1.2. BTL – Gasification/Fischer-Tropsch 6. Feedstocks 6.1.1. Soybean Oil 6.1.2. Palm Oil 6.1.3. Rapeseed Oil 6.1.4. Jatropha 6.1.5. Algae 6.1.6. Animal Fats 6.1.7. Waste Biomass 7. Conclusions 8. Conversion Factors and Calculations 8.1. Conversion Factors 8.2. Calculations 9. References
Thanks for an informative post Robert, there is a lot of missinformation out there on this topic, especially here in Minnesota. My kids were home from school on Thursday and Friday due to cold weather (below -20F plus wind chill) I was sure to tell them that I never got out of school due to cold and I had to walk uphill both ways….
In Minnesota we have many renewable energy mandates. We have wind power mandate laws. A 10% ethanol in gas law and some have been pushing for 20%. I think we have the most E85 stations in the nation. And we are a leader for Biodeisel mandates, but for years we have been dealing with clogged fuel filters. If you understand the blending you will be fine, but most deisel mechanics are not chemists.
Not to worry, we are back in the positve temperatures (after 5 days with negative high temperatures) and the kids will be back to school on Tuesday.
Thank you for the concise and straightforward explaination. A couple of things that I think would add to the article:
What is the net energy return for each of these processes? Cracking and FT are both very energy intensive.
What is the net greenhouse gas emissions for each othese processes? Again, I think that the numbers are very different for each of the processes. A follow on to this would be to discuss net greenhouse gas emissions if non renewable feedstocks are used (petroleum, coal, natural gas) vs renewable (vegetable oil, methane).
A table or graph of these numbers would also be really illustrative.
Benny,I’ve spent more time studying the supply/demand equation,and I’ve concluded the much touted OPEC cuts amount to diddly squat.
Nigeria agreed to a cut of 600,000 bpd. They are so far under quota,they can increase production and still comply. They did increase production in Dec. Venezuela also won’t cut a thing for the same reason. Iran isn’t in compliance with the new quotas. If and when they do choose to comply,they can maintain current oil sales indefinitely,thanks to 15 supertankers full of crude they’ve been unable to sell since April. Add to the mix another 580,000 bpd non-OPEC countries will add to supply this year,and the stage is set for even more glut. Iraq still plans to double production in the next 5 years. That would add another 2.5M bpd to supplies. The Saudi’s are actually producing under quota. They even announced a further cut of 300,000 bpd. I suspect they’re cutting the heaviest,sourest crude,since it’s the hardest to sell in a glutted market. There’s no more storage space,so they either trim production or let it run onto the ground. Spot prices have a ways to fall. Futures prices have a long,long way to fall.
What is the net energy return for each of these processes? Cracking and FT are both very energy intensive.
I got into this some in the book chapter. The energy returns are very dependent upon the feedstock, and there isn’t a lot of experience with feedstocks outside of rapeseed, palm oil, and soybean oil. Most of the others are still more like research projects.
Cheers, RR
I still believe that a nuclear powered CTL plant would be a great option. burning this type of fuel would be similar to a PHEV but instead of putting the nuclear power into a battery you are putting it into the tank.
Maury-
I think so. Making predictions is hard, especially about the future,–Casey Stengel.
The production charts I have seen are breathtaking. Chinese steel production down 20 percent. U.S. auto sales off by one-third. Japanese exports down by a quarter etc. These are the largest reductions in output in the post-WWII era. Worse, they appear to be global.
The global economy is going into recession, just as nearly 10 straight years of crude oil price hikes are generating conservation, and alternative output.
Supertankers are filling up everywhere. Someday, those tankers will have to offload.
I think we will see $10 oil again. Post that on TOD, and you will get censored. I wonder why.
On palm oil: If RR is right about Peak Lite, then there is a great future in palm oil. Yields are rising, and may rise sharply with new hybrids. Palm oil a very profitable crop above $60 a barrel. Brazil could grow square miles of the stuff. We could see several mbd from palm oil alone, in 15-20 years. Mayve more. The trouble is, oil never seems to be able to sustain a high price.
What an excellent post! Thank you for sharing your knowledge for the lay reading public.
I look forward to the book that you will eventually write, even though if it will not come out until you are 50. On an earlier post you asked what would give this work a unique approach. How about energy primer for investors for cutting edge green energy technology. It may give some warning on what approaches could be boondoggles.
As you correctly note, petroleum diesel will gel. It happens here in Japan, and this is not such a cold country, overall. In the winter, the grade of diesel fuel is changed to prevent gelling, but it happens nevertheless to heavy machinery here in the mountains. A remedy used by construction companies is to mix some kerosene into the fuel.
Here is a link to an article for LCA of biodiesel:
http://www.greencarcongress.com/2008/12/new-lca-of-four.html#comments
Unfortunately, there is a charge down loading the paper. I would check to see if they are following ISO 1400 methods and if credit is provided for nitrogen fixing when soy beans are used as a rotational crops.
“Italy’s Eni has announced plans for a facility in Livorno, Italy”.
That’s like calling Rome Roma or Lisbon Lisboa – or Italy Italia. The English language place name of “Livorno” is Leghorn. This is not a quibble as it is important for following up references.
“How about energy primer for investors for cutting edge green energy technology.”
That’s a good idea imo. Lots of reading out there for investers to “make an instant million”,but it’s hard for the average guy to make heads or tails of all the new technology.
How about energy primer for investors for cutting edge green energy technology. It may give some warning on what approaches could be boondoggles.
Better yet: explain the methodology behind seeing through some of these scams. It’s not rocket science.
Better yet: explain the methodology behind seeing through some of these scams. It’s not rocket science.
I agree that would offer more utility. The ‘cutting edge’ could always follow.
RBM
I’ve got a friend that’s a chemical engineer. Smart guy,and well read. But naive as hell. He thinks the government covered up/erased,discombobulated or whatever,the perpetual motion car. Some people fall for penny stocks. Danny falls for millipenny stocks. His last guaranteed winner was .008,but would hit a million gazillion as soon as the world realized how revolutionary the engine was. Danny could use a guide on what’s feasable and what’s not. Heck,that’s half of what Robert does here. Why not make a buck off it?
The English language place name of “Livorno” is Leghorn.
I have actually been there and didn’t know that. I am pretty sure that reference came from an English language release on Eni’s site. But thanks for pointing out the name issue. If they do another edition, I will change it.
In fact, I am going to change it in the essay now.
Cheers, RR
Better yet: explain the methodology behind seeing through some of these scams. It’s not rocket science.
Except sometimes it is. I can listen to a doctor explain to me how they are going to cure cancer within 3 years because of some revolutionary discovery. He can drop all kinds of biomedical nomenclature into the discussion, and I won’t even know which questions to ask.
For instance, let’s say that his studies are done on animal models, and a specific gene is targeted that doesn’t even exist in humans. I don’t know this. He doesn’t mention it because he thinks the technique will work on a gene that does exist in humans. The only thing I can do is listen to an expert with an opposing view, and based on what I do know (and I do know a little biology) – try to figure out which position is more reality-based.
But in general, “if it sounds too good to be true” is a good rule of thumb to start with.
Cheers, RR
The LCA study mentioned in Green Car Congress was done by Argonne National Laboratory. It, or something similar, appears to be available for free at
http://www.transportation.anl.gov/pdfs/AF/467.pdf
RR, what do you think of Argonne’s GREET model? Do you think it’s at all close to reality?
Nevermind. I think I found the answer to that in
http://i-r-squared.blogspot.com/search/label/Michael%20Wang
I just checked google’s language translation tools. It turns out that it knows enough to translate Livorno as Leghorn and vice versa, so probably the reference problem won’t be a big deal for google searches.
Thanks for the link Clee. Very good LCA. Clearly Indiana farmers processing the energy out of the animal feed they are already growing is a good environmental choice.
It is always easy to spot the agenda driven. What about land use in Brazil? LCA is about making choice to do what is already being done. Why do people always bring up Brazil when we are talking about Indiana? I think it would be rather odd tor Argonne labs to study Brazil. I have the same problem professors from Cornell and Berkley studying the midwest. Clearly, California and New York have enough issue to deal with. An of course, there are the Texas and Oklahoma oil interests explaining why Indiana farmers should not produce a petroleum substitute when they are no longer capable of meeting domestic needs.
“In recent years, the sales volume for biodiesel in the United States has increased dramatically: from about 2 million gallons in 2000, to 75 million gallons in 2005, to 250 million gallons in 2006 (National
Biodiesel Board 2007).”
Good job on the exponential growth President Bush. It was nice to have a president that did more than talk about renewable energy. Will the new ‘change’ president order the helmsman to steer 270 or order steady as you go. Doubling the amount of renewable energy in three year will be a challenge for the dems that historically has been their record. I do expect a share increase in studies of social justice.
It is always easy to spot the agenda driven. What about land use in Brazil? LCA is about making choice to do what is already being done. Why do people always bring up Brazil when we are talking about Indiana?
You are correct if the LCA is specifically defined as soybean-derived biodiesel of a specified amount being produced in Indiana. But as you should know, LCAs are very situation dependent.
For instance, multiply the amount of biodiesel produced in Indiana by 10. Does the LCA change? Almost certainly. Massively scale up the usage of biofuels. Is the LCA for some specific amount of a specific biofuel the same as for a greatly increased amount? Of course not. So it may very well be appropriate to discuss Brazil in the context of an LCA. It depends on what the parameters are.
RR
Hey Robert,
Great post, as usual. There appears to be a typo in the first paragraph in the Biodiesel Characteristics section, unless I’m just reading it wrong.
The third sentence states, “Compared to petroleum diesel, a pure blend of biodiesel was estimated to increase the emission of NOx by 10%…” The sixth sentences then says, “While confirming the NOx reduction observed in the EPA studies…”
Me: It’s not rocket science.
RR: Except sometimes it is.
Agreed, some of it gets pretty technical. Still, I was thinking along the lines of basic mass and energy balances. And the Laws of Thermodynamics: Put 1 kWh in, and you are going to get LESS that 1 kWh of useful energy out. The thing that kills all hydrogen schemes.
Take the way you picked apart TDP with minimal reference to the technical details. Your main argument, as I recall, was that scaling up from bench scale to full scale was fraught with unpleasant discoveries, something CWT now fully appreciates. Your contention that cost estimates based on bench scale data are rough order of magnitude estimates at best, was proven to be tragically accurate…
Darn it, Maury, if your friend is that qualified and he STILL believes in perpetual motion, well, there is nothing to be done for people like that. That’s got to be a small minority, though!
Good job on the exponential growth President Bush.
That’s amazing, Kit. I mean after all that Mr. Bush still has supporters out there. Congratulations on being part of that exclusive a club. And, no, we’re not interested in the interesting debate you had at your last meeting…
And, oh yeah, burning food is a great solution to our energy problems – as long as we can figure out a way to stop eating. And I’m sure if campaign donors made a pile of money out of this scheme it is just an unfortunate coincidence. Like many others during the last eight years…
“And, oh yeah, burning food is a great solution to our energy problems “
I love the idea. Let’s find out now if we can feed 9 billion people,the expected population by 2050,by feeding 6 billion people and 3 billion cars.
We can even put those cars on a low calorie diet. Feed ’em wood chips or chicken feed/corn.
Green Crude by David Ewing Duncan at Potfolio.com on 01-07-09
Author regurgitates spin of companies looking for investors.
RR is correct that that biodiesel may not scale up for a sustainable solution that is a significant share of the market. We are a long way from running out of integrated holistic solutions for environmental problems. For those who worry about trivial issues like AGW and peak oil, I give you peak phosphorous and all the environmental issues associated with mining, processing and use. This LCA only briefly touched on the issue.
I would be interested in those who think wind and solar powering BEV are integrated holistic solutions.
“And, oh yeah, burning food is a great solution to our energy problems …”
The LCA discussed this. The protein value of soy beans is not lost in the soy meal.
One of the local AGW zealots who likes to chain himself to the fence of power plants thinks the solution is burning corn in a pellet stove. I was checking air quality today at AIRNOW and surprised to see a warning for the Seattle area. Wood smoke from home heating.
Here's an interesting story about biodiesel made from tallow.
From
http://www.abc.net.au/rn/scienceshow/stories/2008/2441773.htm#transcript
– Rod
Transcript
Robyn Williams: If you boil up the carcasses of cattle or sheep you get tallow, and that could be a prime source of fuels. It's an industry potentially worth billions if you can keep the fluids liquid, even at low temperatures. Here's Stephen Clarke at Flinders University in South Australia.
Stephen Clarke: Tallow is basically the fat that comes out of the rendering process of meat and sheep when it's processed by the abattoirs.
Robyn Williams: And what normally happens to it?
Stephen Clarke: In the past it's widely been used in a variety of applications; making soap and cosmetics. But that use has declined and the price of tallow has dropped, so the meat industry was looking for alternative options, and converting the tallow into biodiesel was a good opportunity. Most of the biodiesel plants in Australia actually use tallow as a feedstock.
Robyn Williams: And if they couldn't sell it to biodiesel people, were they turfing it?
Stephen Clarke: No, they were still selling it but the price had dropped to $400 a tonne which was a very low price, and at that price it also made conversion into biodiesel a good option because fuel was selling for around $1.00 – $1.10 a litre at that time when it was at that price.
Robyn Williams: And I suppose the advantage is you've got it all in one place so you don't have to transport it gigantic distances, and you get sufficient supply and constant supply so you can provide for an industry.
Stephen Clarke: That's right. There's actually enough tallow produced in Australia to meet about 10% of Australia's diesel needs if it was all converted into biodiesel.
Robyn Williams: And converting it is the problem, isn't it, because at low temperature it's solid.
Stephen Clarke: Yes, if you make what's called B100 biodiesel out of tallow, which is the 100% biodiesel, it has a freezing temperature of between 10°C and 15°C, depending on the actual chemistry make-up of that feedstock. This is the main reason why tallow based biodiesel is blended into normal diesel and sold as maybe B5 or B10. We developed a technology that was able to push the freezing temperature of that blend from about 1°C down to about -6°C, and this meant that the properties of the biodiesel blend was very similar to normal diesel that's used in Australia in winter conditions.
Robyn Williams: So you don't want the fuel in your tank going solid like lard because that would be counterproductive.
Stephen Clarke: Yes, it basically turns into a waxy solid and it would freeze up your tank, so you obviously don't want to have that in the middle of winter.
Robyn Williams: Of course that's one of the main problems with getting a new fuel for aircraft because at high altitudes it's even lower, it's something like -40°C, and that's why kero is the only option so far.
Stephen Clarke: Yes, that's right. We've also been working in that area with a different type of B100 biodiesel and we've been able to push the freezing temperature of that down to -55°C, and the specification for jet fuel, what's called jet A1 fuel, is -47°C. So we think we've got something that's quite promising.
Robyn Williams: That's very interesting because Richard Branson has been talking about that for his aircraft so that you can get biodiesel which is reliable and stable to use on a constant basis, because you don't want your fuel to fail up there in the sky.
Stephen Clarke: No, that's right. One of the fuels that's been used in that area is made from what's called Jatropha which is a generation-two feedstock. On its own it's not suitable and you can't even convert that into biodiesel, it can be used in jet fuels, but in America they've developed a technology where they hydrogenate the Jatropha and then they put it through a normal refining process. They basically convert the Jatropha oil to a normal petroleum type product, and then they distil it and extract the jet fuel type component. Air New Zealand and Boeing are doing trials on that material at the moment, they're doing in a 50-50 blend.
Robyn Williams: Jatropha is in fact a tree that grows very nicely in Australia in the wastelands.
Stephen Clarke: Yes, that's right. One of the issues with Jatropha is that some of the state governments class it as a noxious weed so we actually can't grow it in Australia, yet it's being grown all around the world in India, Africa and some of the Asian countries as a feedstock for biodiesel.
Robyn Williams: Yes, but some different species grow wild as well, don't they, here?
Stephen Clarke: Yes, I think the Australian government is concerned about it growing out of control.
Robyn Williams: Okay, here you are looking at the future of biodiesel and you're working from Flinders University and trying to lower temperatures. Are you able to give me the secret?
Stephen Clarke: I can't tell you how we lower the temperature but essentially the oil is converted by what's called a transesterification reaction, it is reacted with a base catalyst and methanol, and for every ten litres of tallow or starting material you start with, you end up with ten litres of biodiesel, and you end up with one litre of glycerol as a by-product. Unfortunately in Australia most of them were burning that by-product, some were converting it into pharmaceutical grade glycerine, but you can actually use that as a platform chemical to make a whole range of new chemicals like plastics and pharmaceutical drugs and so on.
And if you consider…we use 15 billion litres of diesel in Australia. If we were to replace that with 15 billion litres of biodiesel, we would be producing 1.5 billion litres of glycerol as a by-product. You can use this to be producing value added products, maybe selling at $10 or $20 a litre, that underpins the operation of the biofuels industry. And this is exactly what the petroleum industry has been doing. About 15% out of every barrel of crude goes towards petrochemicals, which are high profit, high value or value added products that underpin the sale of fuel in the petroleum fuel industry. Yet the biofuels people in Australia and around the world don't do that, they just focus on trying to produce a low profit margin, high volume product. And that's why the industry struck troubles last year and essentially the Australian biodiesel industry collapsed because prices of feedstock went up and they didn't have the buffer of what we call a value added bio-refinery industry to buffet the…
Robyn Williams: And there are only about two plants going at the moment.
Stephen Clarke: Two major plants going in Australia, yes.
Robyn Williams: Where are they?
Stephen Clarke: One's in Victoria and I think one's near Albury.
Robyn Williams: So the second question about biofuels, apart from the chemistry, the secret of which you'll tell me one day quietly but we've got the principle; is a reliable supply not conflicting with arable land and growing food. So what you're looking at is stuff that is waste that is reliable and in substantial supply, and you're saying that that can be the basis for a substantial biofuel industry in Australia.
Stephen Clarke: Yes, that's one component of it. The other component is the agricultural feedstocks for making biofuels are called generation-one or gen-one feedstock. There's also a strong push to move towards the gen-two or generation-two feedstocks, and micro-algae is seen as an opportunity. Micro-algae has the opportunity of providing lipids or oils or fats for biodiesel. They can also produce starch for ethanol production, and in some cases you can even produce hydrogen, so for the hydrogen economy, the generation-three cars, the battery cars that we've been hearing so much about.
And the beauty with the generation-two micro-algae processes is that the land needed is about one-thirtieth of the land that you need for generation-one biofuels. To give you an example, if America was to produce all of its ethanol from corn production, they would need 8.5 times the area of crop land that they use at the moment just to meet their fuel requirements, which obviously is not feasible. So you've got to look at alternatives, and the generation-two feedstocks like cellulosic feedstocks and the micro-algae feedstocks provide you that opportunity to get the quantity. At the moment the price of those fuels are too expensive, so it needs a lot of research to help drive the cost down to make it feasible. But once that's achieved then basically we can overcome the problems of petroleum fuels.
Robyn Williams: So in five or ten years time do you expect biofuels to have taken off, if people have got good sense?
Stephen Clarke: Yes, I think that's the sort of timeframe that we're looking at is probably five to ten years. Typically the price of oil coming from micro-algae at the stage…and the technology is there, it works, it's just the price is around $5 a litre, which obviously when you're competing against crude at 30c – 40c a litre is not the best. It's a bit like the colour television; when that first came out it was selling for $2,000, $3,000 a set, and now we can buy colour televisions for $300 or $400. So as we develop the technology and improve the processes I think the prices will come down and it should be competitive with petroleum fuels.
Robyn Williams: Dr Stephen Clarke is leader of the biofuels group at Flinders University in Adelaide, and he estimates the industry could be worth $50 billion a year to Australia.
Guests
Stephen Clarke
Leader Material and Bioenergy Group School of Chemistry, Physics & Earth Sciences Flinders University Adelaide South Australia
http://www.scieng.flinders.edu.au/cpes/people/clarke_s/index.html
Presenter
Robyn Williams
Producer
David Fisher
ROBERT–
don’t wait for book publication to make money. sell selected parts to gov’t entities[Minn. eg.] to keep the public safe from illiterate POLITICIANS/BEAUROCRATS.
i have a strong suspicion the highway depts and farmers know the differences among diesel offerings.
your reference list may be missing RENTECH as FT source[ala Syntroleum]. RTK has only operating pilot line in USA[10 bbl/day].
fran
There appears to be a typo in the first paragraph in the Biodiesel Characteristics section, unless I’m just reading it wrong.
I am in Europe, and don’t have the book with me. When I return, I am going to pull the studies, but it appears to me that you are correct. I think ‘reduction’ should say ‘results.’
I must have read that chapter a hundred times, and an editor read it several times. Hard to believe that things still slip through. Thanks for catching it.
Cheers, Robert
Author regurgitates spin of companies looking for investors.
I love how people always throw out Bill Gates as an endorsement. I guess people don’t realize that his last foray into biofuels (Pacific Ethanol) resulted in an 80 to 90% loss of his investment.
I will reiterate. If the topic is software, or things computer-related, I care about what Bill Gates has to say. If the topic is biofuels, he doesn’t bring any special knowledge to the table. He is a layman in that area, and as he found out, laymen can be had.
RR
“I’ve got a friend that’s a chemical engineer. He thinks the government covered up/erased,discombobulated or whatever,the perpetual motion car.”
Maury~
An engineer who thinks perpetual motion is possible wasn’t paying attention in his thermodynamics courses.
“It’s not rocket science.”
Optimist ~
Small point perhaps, but the correct phrase should be, “It’s not rocket engineering.”
Getting to the Moon wasn’t a question of understanding or discovering the science. The science was well understood. It was a matter of engineering the materials and technology to put the science into reality.
clarification on gasification vs. combustion:
You state
“When an organic material is burned (e.g., natural gas, coal, biomass), it can be completely oxidized (gasified) to carbon dioxide and water..”
do you mean combusted instead of gasified? I thought “gasified” meant partial combustion.
neel
Robert – please update your blog to remove reference to the Minnesota school bus issue, which was NOT caused by biodiesel.
http://nbb.grassroots.com/resources/BloomingtonBusReport.pdf is the final report from the independent analysis. Every bus was a specific make and model, which used a different housing for the fuel filter, exposing it to far more cold than the others (among other things).